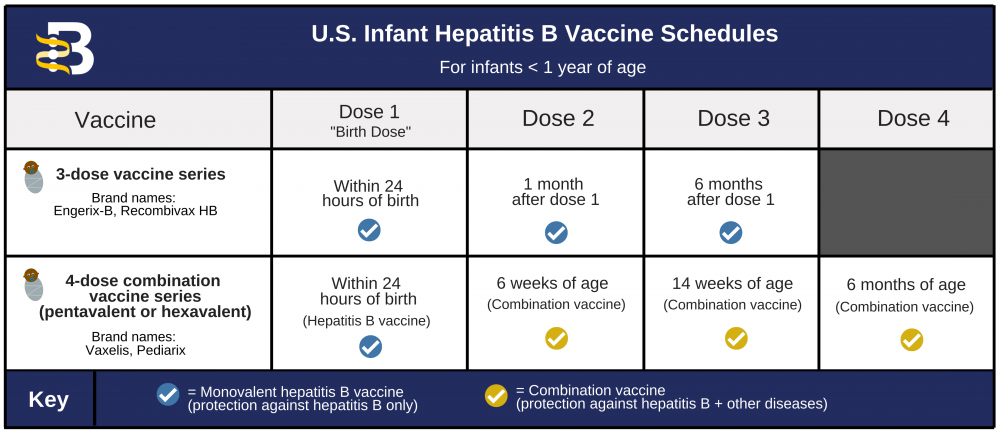
Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials - The Lancet

Inhibition of Complement Factor 3 in Geographic Atrophy with NGM621: Phase 1 Dose-Escalation Study Results - American Journal of Ophthalmology
Should FDA Reconsider Allowing the Pooling of COVID-19 Vaccine Doses to Obtain Additional Doses? | Institute For Safe Medication Practices
Walgreens Tioconazole Ointment 6.5 Percent, 1-Dose Treatment for Vaginal Yeast Infection | Walgreens

A Randomized Trial Comparing the Safety, Adherence, and Pharmacodynamics Profiles of Two Doses of Sodium Bicarbonate in CKD: the BASE Pilot Trial | American Society of Nephrology
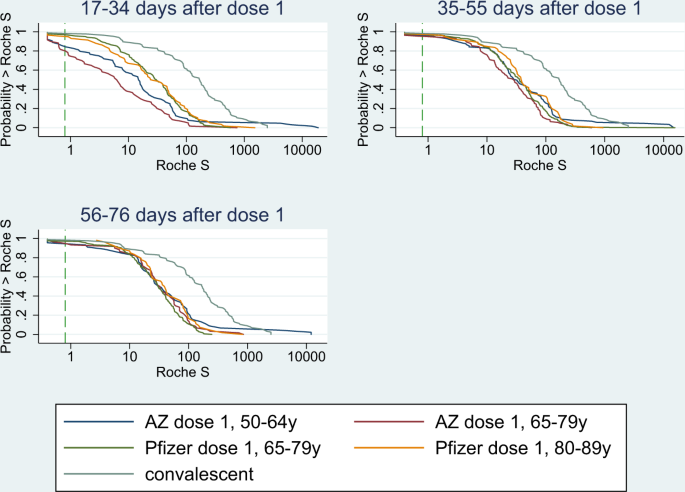
Serological responses and vaccine effectiveness for extended COVID-19 vaccine schedules in England | Nature Communications

Effectiveness of a fourth dose of mRNA COVID-19 vaccine against all-cause mortality in long-term care facility residents and in the oldest old: A nationwide, retrospective cohort study in Sweden - The Lancet

Public health impact of delaying second dose of BNT162b2 or mRNA-1273 covid-19 vaccine: simulation agent based modeling study | The BMJ
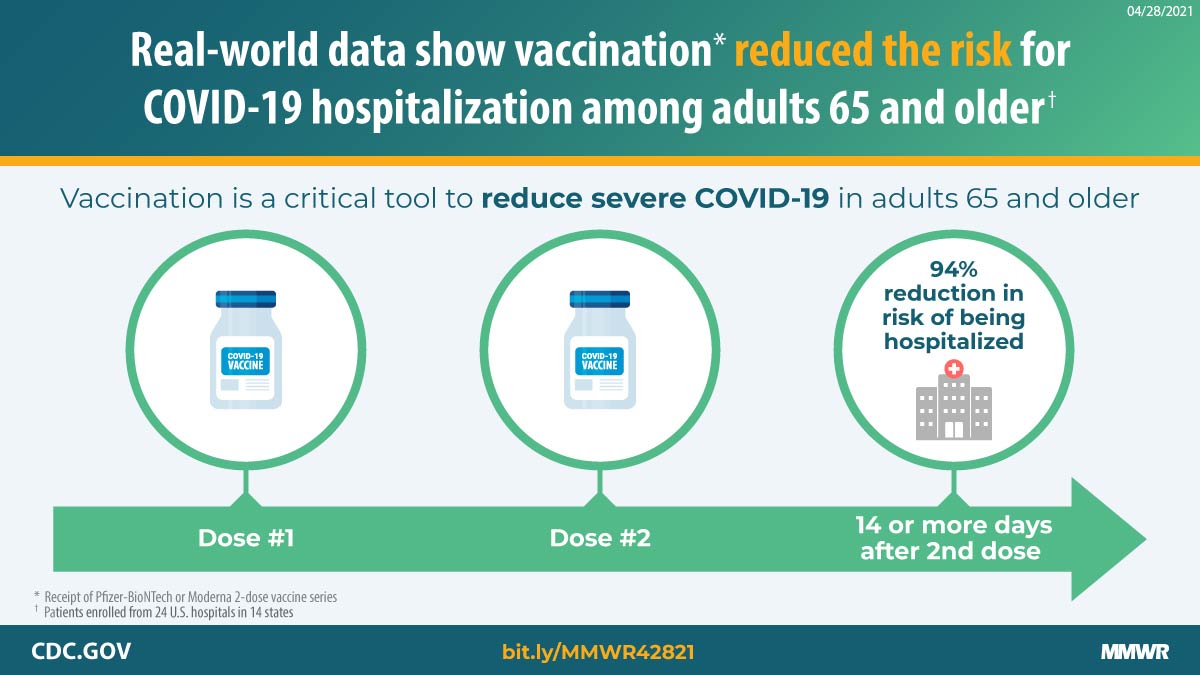
Effectiveness of Pfizer-BioNTech and Moderna Vaccines Against COVID-19 Among Hospitalized Adults Aged ≥65 Years — United States, January–March 2021 | MMWR

Effectiveness of a third dose of BNT162b2 mRNA COVID-19 vaccine in a large US health system: A retrospective cohort study - The Lancet Regional Health – Americas

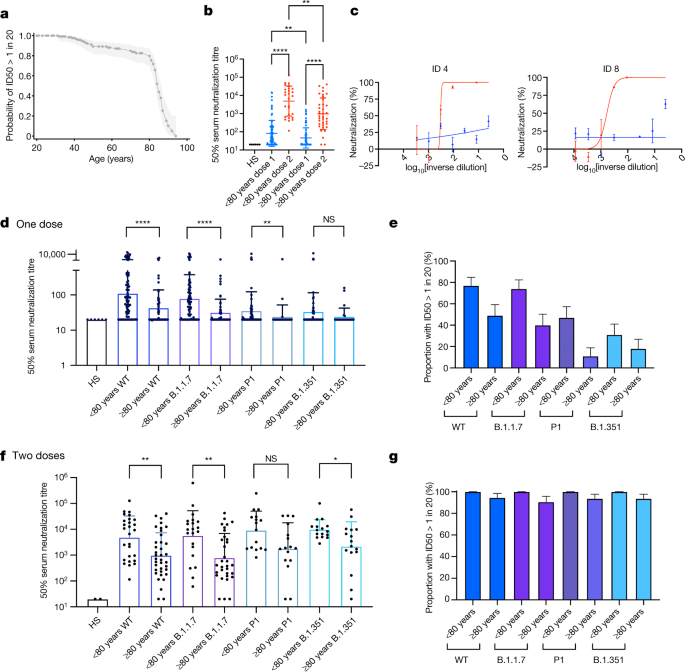
Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity - ScienceDirect










![Calculation of Equivalent Dose and Effective Dose [MOE] Calculation of Equivalent Dose and Effective Dose [MOE]](https://www.env.go.jp/en/chemi/rhm/basic-info/1st/img/img-02-03-06.png)