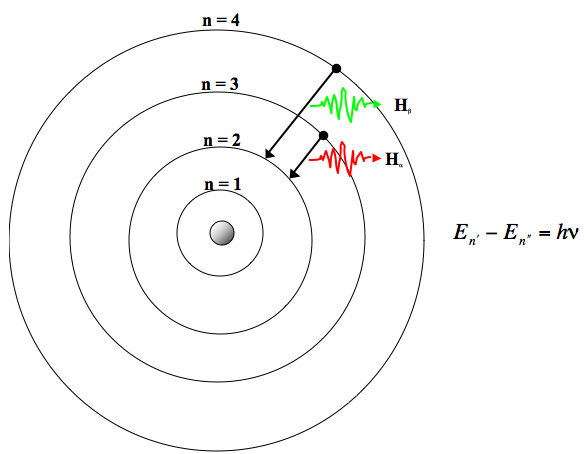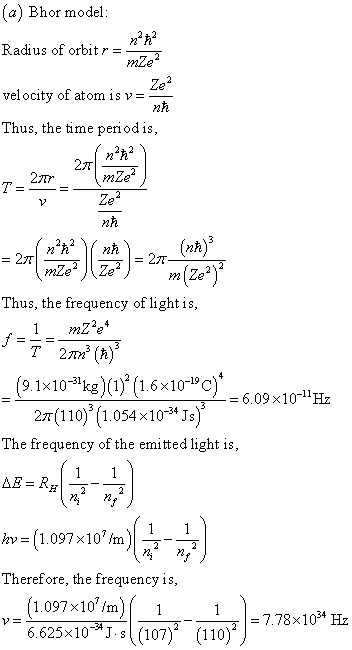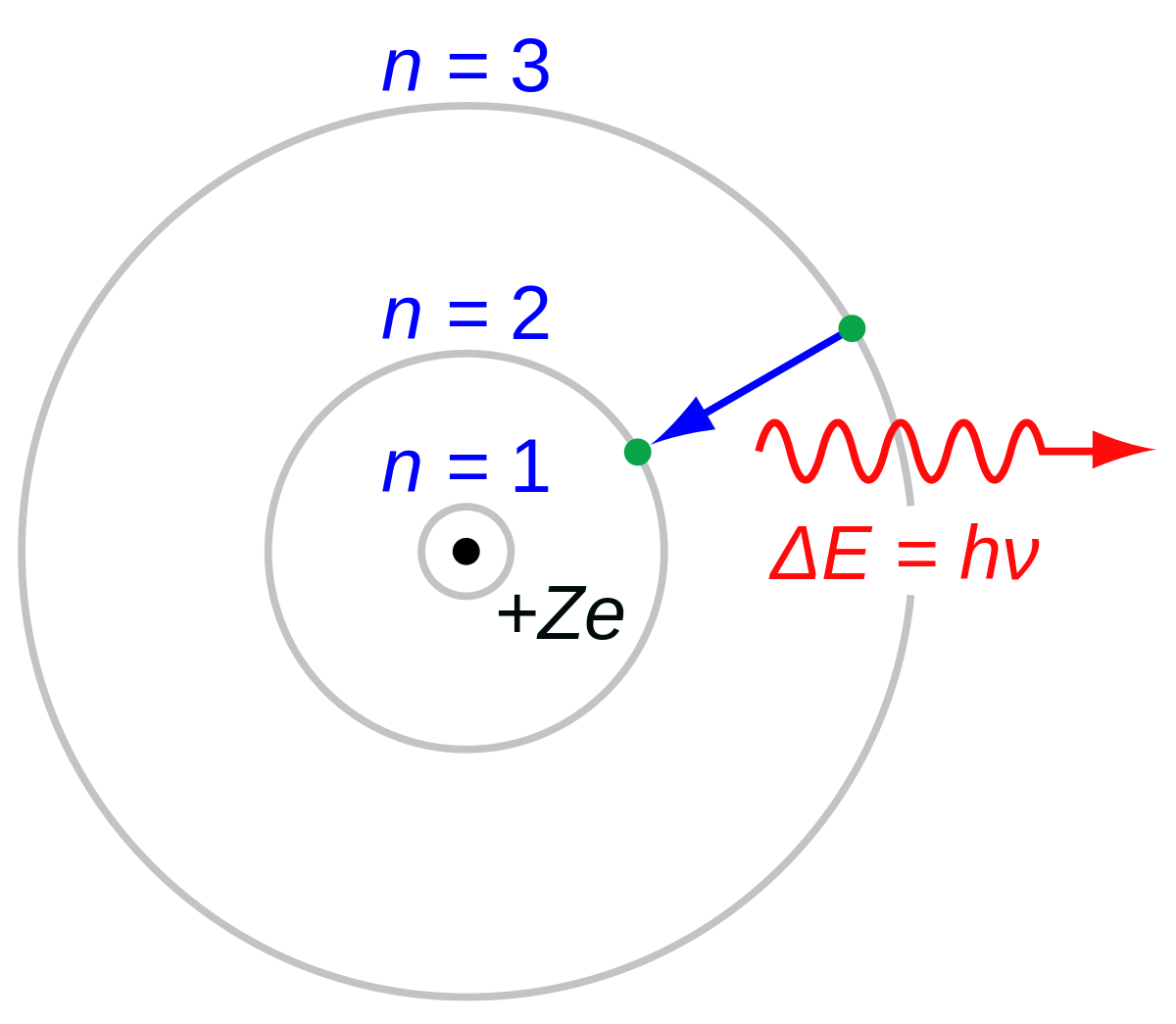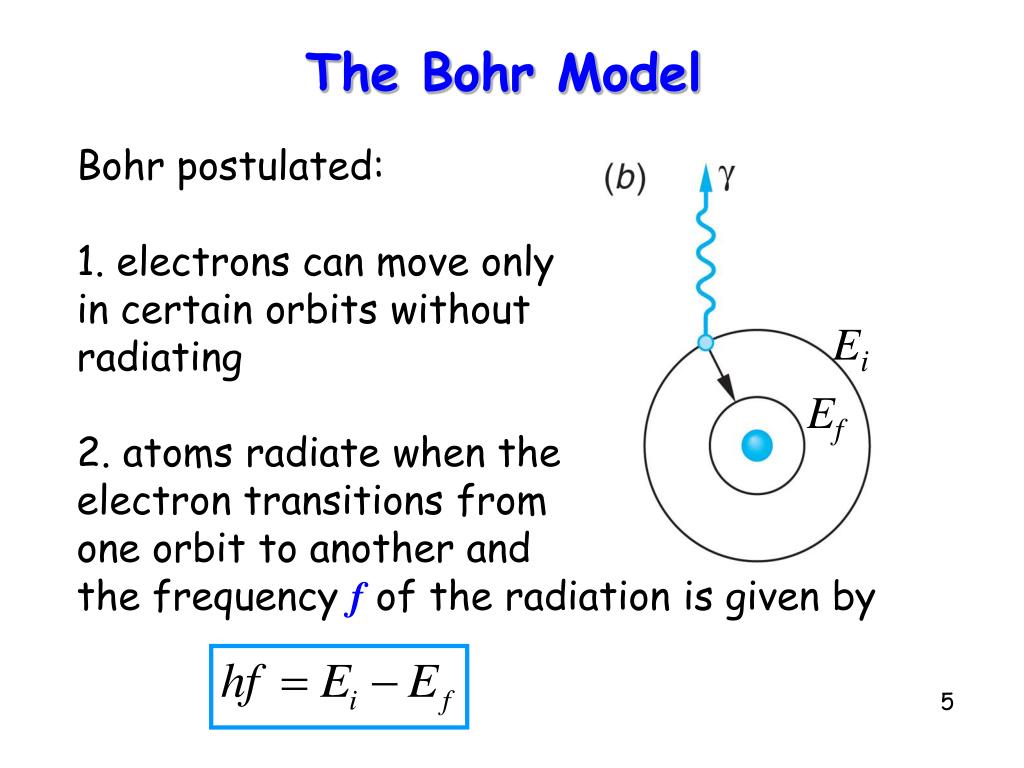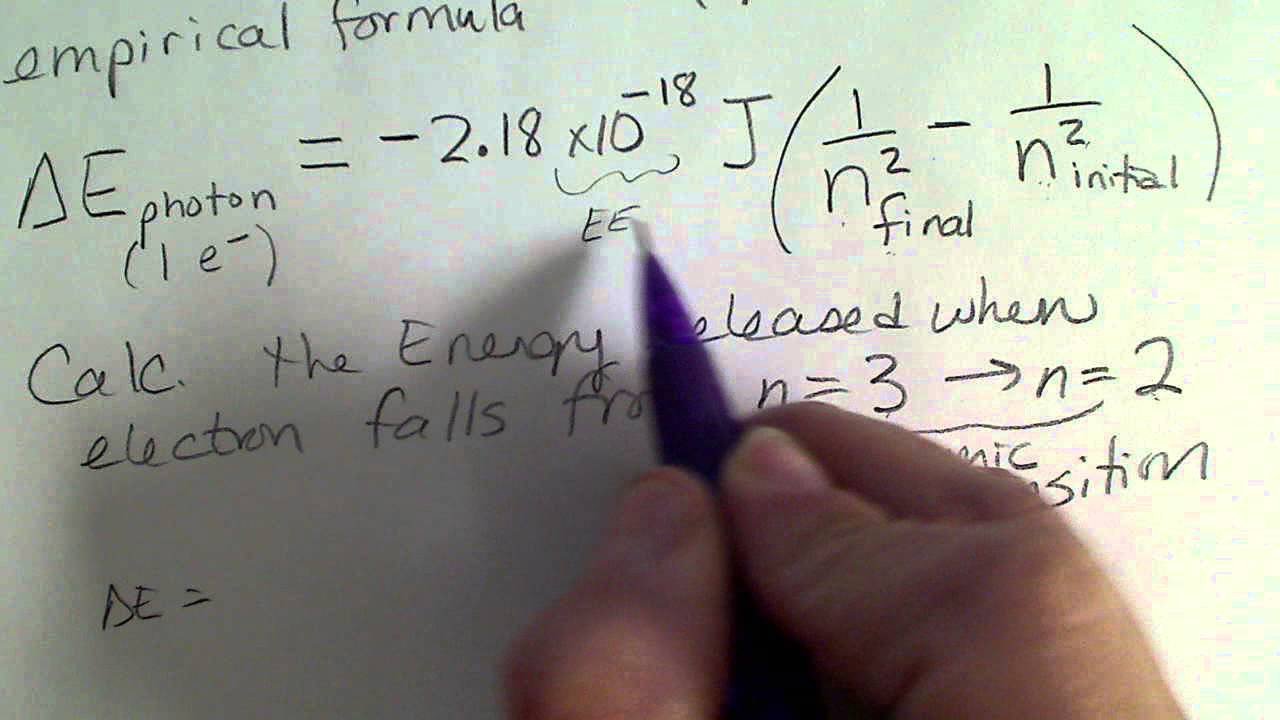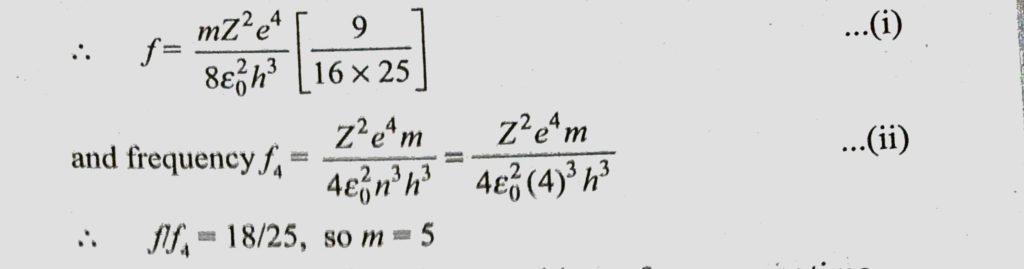
A Bohr hydrogen atom undergoes a transition n = 5 to n = 4 and emits a photon of frequency f. Frequency of circular motion of electron in n = 4 orbit

What is the frequency of revolution of electron present in `2nd` Bohr\'s orbit of `H-` atom ? - YouTube
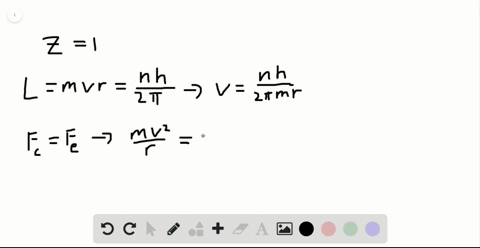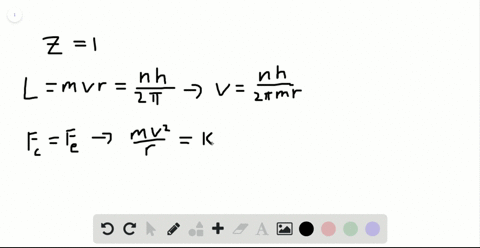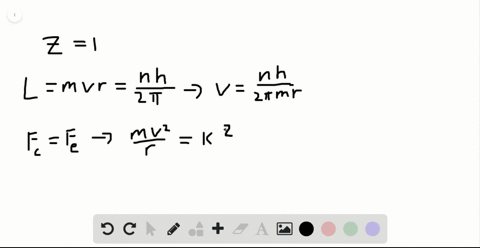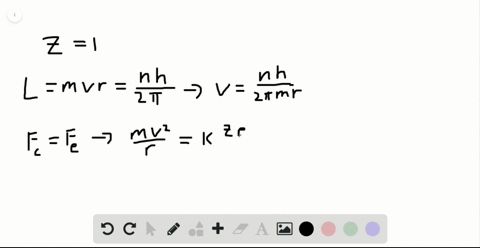
SOLVED:Determine the speed and frequency of an electron moving around the first Bohr orbit in hydrogen. According to classical physics, the atom should emit electromagnetic radiation at this frequency. In what portion

If the velocity of electron in the first Bhors orbit of H atoms is s 2.19 × 10^6m/s .calculate the velocity of electron in Bohrs second orbit?

The frequency of radiation absorbed or emitted when transition occurs between two stationary states that differ in energy by: Δ E , is given by: v = ΔEh = E2-E1h Where E1

SOLVED: Using the Bohr model of the Hydrogen atom, what is the frequency of the photon released when an electron drops from the n = 4 level to the n = 2

Calculate frequency of revolution of electrons in `4^(th)` Bohr orbit of `Be^(+3)` ion. Given that - YouTube