Repeated-doses and reproductive toxicity studies of the monoterpene 1,8-cineole (eucalyptol) in Wistar rats - ScienceDirect

In Silico Models for Repeated-Dose Toxicity (RDT): Prediction of the No Observed Adverse Effect Level (NOAEL) and Lowest Observed Adverse Effect Level (LOAEL) for Drugs | SpringerLink

Analysis of repeated dose toxicity studies. The study protocols and... | Download Scientific Diagram
![PDF] Read-across of 90-day rat oral repeated-dose toxicity: A case study for selected β-olefinic alcohols | Semantic Scholar PDF] Read-across of 90-day rat oral repeated-dose toxicity: A case study for selected β-olefinic alcohols | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/23f7da37b5c0cbba3d05a372ce6a08ccaa534203/9-Table1-1.png)
PDF] Read-across of 90-day rat oral repeated-dose toxicity: A case study for selected β-olefinic alcohols | Semantic Scholar

Read-across of 90-day rodent repeated-dose toxicity: A case study for selected simple aryl alcohol alkyl carboxylic acid esters - ScienceDirect
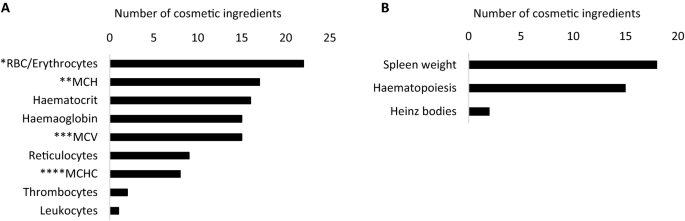
Screening of repeated dose toxicity data in safety evaluation reports of cosmetic ingredients issued by the Scientific Committee on Consumer Safety between 2009 and 2019 | SpringerLink

Identification of repeated dose toxicity studies described in 88 safety... | Download Scientific Diagram

Foods | Free Full-Text | Single and Repeated Dose 28-Day and 13-Week Toxicity Studies of Oil Prepared from the Internal Organs of the Japanese Giant Scallop (Patinopecten yessoensis) in Mice
Preliminary Assessment of Acute and 28-Day Repeated Dose Oral Toxicity of a Newly Developed Herbal Mixture on Experimental Anima
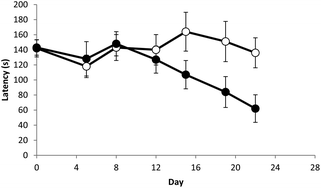
Functional assessments in repeat-dose toxicity studies: the art of the possible - Toxicology Research (RSC Publishing)
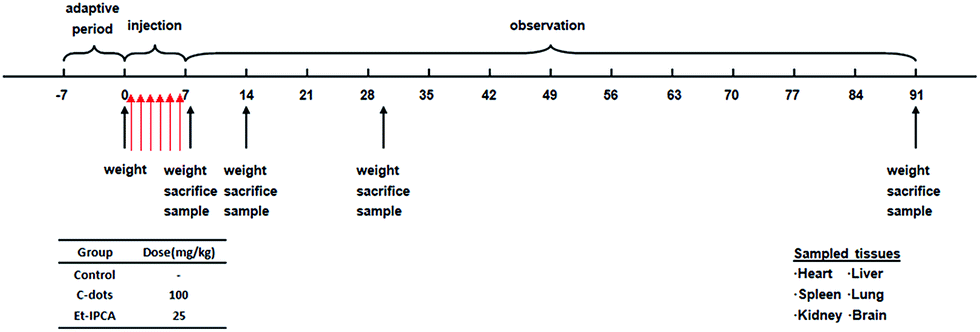
Single and repeated dose toxicity of citric acid-based carbon dots and a derivative in mice - RSC Advances (RSC Publishing) DOI:10.1039/C5RA18391J

Safety evaluation of aqueous extracts of Sanghuangporus vaninii fruiting body in Sprague–Dawley rats - Huo - 2020 - Food Science & Nutrition - Wiley Online Library
![PDF] Thesaurus for histopathological findings in publically available reports of repeated-dose oral toxicity studies in rats for 156 chemicals. | Semantic Scholar PDF] Thesaurus for histopathological findings in publically available reports of repeated-dose oral toxicity studies in rats for 156 chemicals. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b71f8ced3d604b24ef24fee271c2ea58800a2f3c/2-Table1-1.png)
PDF] Thesaurus for histopathological findings in publically available reports of repeated-dose oral toxicity studies in rats for 156 chemicals. | Semantic Scholar
Description of prototype modes-of-action related to repeated dose toxicity - Publications Office of the EU
Preclinical toxicity and pharmacokinetics of a new orally bioavailable flubendazole formulation and the impact for clinical trials and risk/benefit to patients | PLOS Neglected Tropical Diseases

180 Day Repeated-Dose Toxicity Study on Forchlorfenuron in Sprague–Dawley Rats and Its Effects on the Production of Steroid Hormones | Journal of Agricultural and Food Chemistry